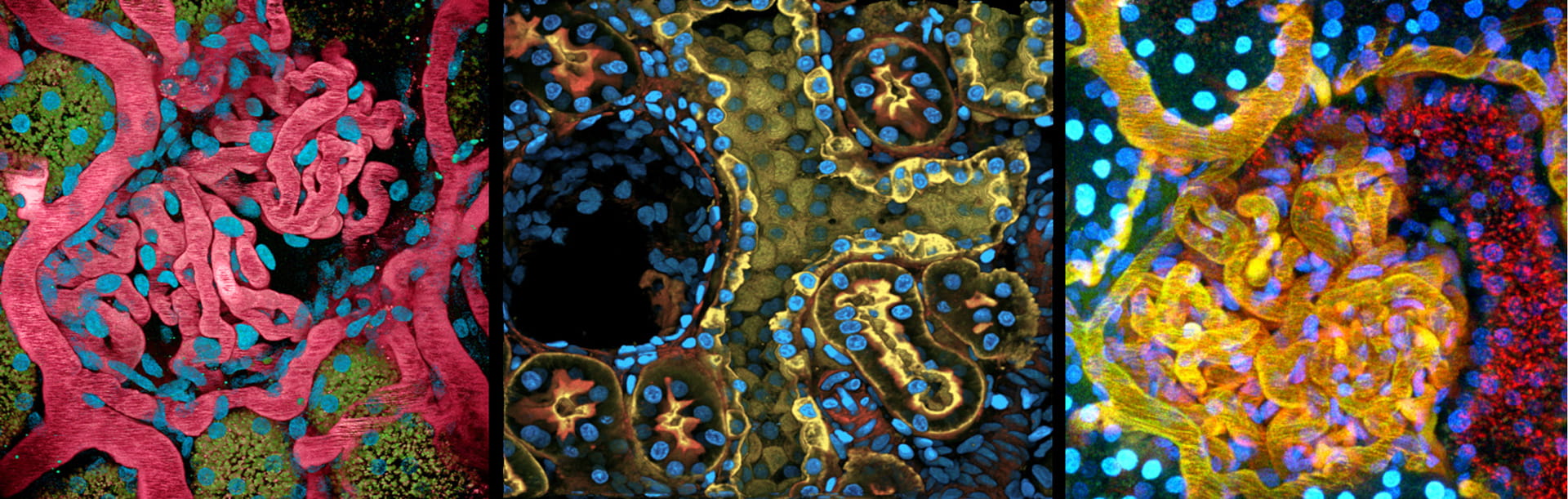
The Indiana University George M. O’Brien Center has, for the last 20 years, developed a unique, highly interactive, integrated and synergistic team of physicians, basic scientists, computer scientists, engineers and a highly trained and committed technical support team to provide local, national and international users cutting edge intravital optical microscopy and 3-dimensional quantitative digital image analysis of the kidney. The development of user-based customization of specialized techniques of microscopy and quantitative analysis has been the hallmark of our NIH P-30 Center.
The newest addition to this center is a quantitative 3-dimensional tissue imaging core to address the need of specialized expertise and infrastructure to support the collection and quantitative analysis, large scale, high content and high resolution microscopy data. Therefore, our center provides renal/urological researchers with a novel set of research methods, quantitative analysis tools and fluorescent biosensors or probes not available elsewhere to facilitate biomedical research, drug discovery and therapeutic approaches to kidney and urologic diseases.